Global Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market to Reach USD 13.3 billion by 2036, Driven by Decentralized Trials and Patient-Centric Delivery Models
 clinical trial logistics and direct-to-patient delivery platforms market is moderately fragmented, with competition defined by global network capabilities, cold-chain validation, and integration with clinical systems.
clinical trial logistics and direct-to-patient delivery platforms market is moderately fragmented, with competition defined by global network capabilities, cold-chain validation, and integration with clinical systems.
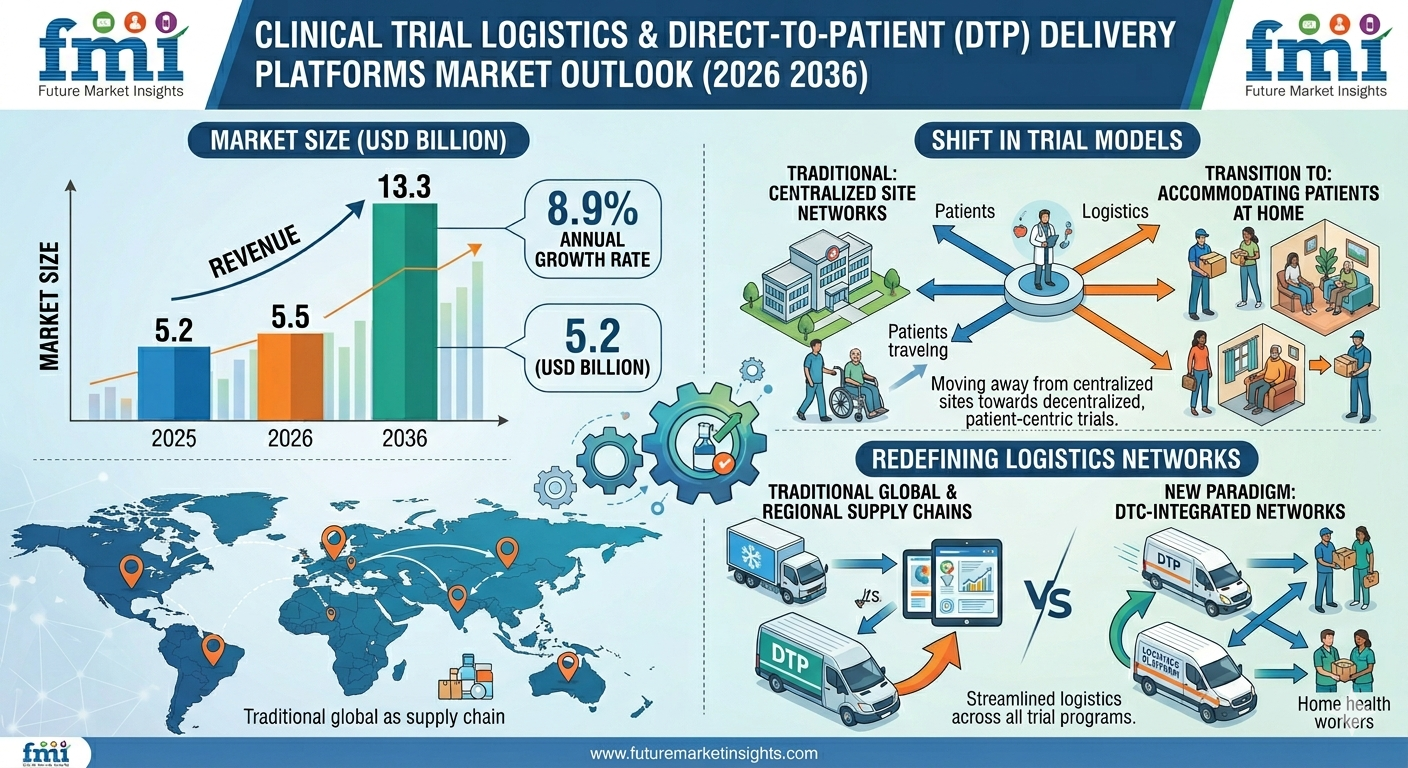
NEWARK, DE / ACCESS Newswire / April 6, 2026 / The clinical trial logistics and direct-to-patient delivery platforms market stood at USD 5.2 billion in 2025 and is projected to reach USD 5.5 billion in 2026, reflecting an annual growth rate of 8.9%. Total revenue is expected to reach USD 13.3 billion by 2036. according to Future Market Insights (FMI). This expansion is fueled by the rapid shift toward decentralized and hybrid clinical trial models, enabling patient-centric approaches and redefining global trial execution frameworks.
The market is evolving as clinical operations transition away from traditional site-based models toward flexible systems that incorporate home-based care, direct drug delivery, and remote patient monitoring. These advancements are reshaping logistics networks, requiring precision-driven coordination, temperature-controlled supply chains, and real-time visibility across trial ecosystems.
Get Access of Report Sample: https://www.futuremarketinsights.com/reports/sample/rep-gb-32583
Clinical trial logistics and direct-to-patient delivery platforms market Quick Stats Snapshot
2026 Market Value: USD 5.5 billion
2036 Forecast Value: USD 13.3 billion
CAGR (2026-2036): 8.9%
Leading Trial Model: Hybrid Trials (42.6% share)
Leading Temperature Segment: Cold Chain (47.2% share)
Top End User: Biopharma Sponsors (49.5% share)
Market Momentum: Rise of decentralized trials and patient-centric logistics
Market Momentum: Decentralization and Patient-Centricity Driving Growth
The increasing complexity of clinical trial protocols and the need to improve patient recruitment and retention are accelerating the adoption of direct-to-patient (DTP) logistics models. By minimizing patient travel and enabling home-based treatment, sponsors are enhancing trial participation while maintaining protocol integrity.
Advanced logistics solutions now integrate temperature-controlled transport, real-time tracking, and automated routing systems to ensure timely and compliant delivery of investigational products. These capabilities are essential for supporting biologics, gene therapies, and other temperature-sensitive treatments.
Key Market Dynamics
Rising adoption of decentralized and hybrid clinical trial models
Increasing demand for patient-centric delivery systems
Growth in biologics and temperature-sensitive therapies
Regulatory support for remote trial execution
Integration of digital platforms with logistics operations
Market Segment Insights
Service Type Leadership
Transport management leads the segment with a 31.8% share, reflecting the critical importance of precise scheduling, routing, and coordination in decentralized trial execution.
Trial Model Insights
Hybrid trials dominate with a 42.6% share, as sponsors combine in-clinic assessments with remote patient engagement to balance oversight and convenience.
Delivery Mode Insights
Depot-to-patient delivery leads due to centralized inventory control and improved consistency across global trial operations.
Regional Insights
North America: Leads with advanced clinical infrastructure and strong regulatory clarity supporting decentralized trials
Europe: Growth driven by strict compliance standards and emphasis on data integrity and chain-of-custody
Asia-Pacific: Fastest-growing region, with India (10.8% CAGR) and China (10.2%) leading due to expanding trial volumes and digitalization
Competitive Landscape: Focus on Network Strength and Compliance
The market is moderately fragmented, with competition defined by global network capabilities, cold-chain validation, and integration with clinical systems. Key players include:
Marken
World Courier
Thermo Fisher Scientific
DHL Group
Almac Group
PCI Pharma Services
Parexel
These companies are investing in predictive routing, sensor-enabled tracking, and platform-based orchestration to enhance delivery precision and regulatory compliance.
Market Drivers and Challenges
Growth Drivers:
Increasing protocol complexity in clinical trials
Rising demand for patient-centric trial models
Expansion of biologics and advanced therapies
Regulatory alignment supporting decentralized trials
Challenges:
Cross-border regulatory fragmentation
Cold-chain logistics complexity
Reverse logistics for biological samples
Integration gaps between digital and physical systems
Strategic Takeaways for Decision Makers
Invest in direct-to-patient logistics infrastructure
Strengthen cold-chain and temperature monitoring capabilities
Leverage AI for predictive routing and delivery optimization
Expand hybrid trial capabilities to improve patient retention
Ensure compliance through robust chain-of-custody systems
For a comprehensive understanding of the latest formulation trends and to explore the full strategic insights on Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market, please visit the official report page: https://www.futuremarketinsights.com/reports/clinical-trial-logistics-and-direct-to-patient-delivery-platforms-market
Why This Market Matters Now
The shift toward decentralized clinical trials is fundamentally transforming the pharmaceutical research landscape. By enabling broader patient access, improving retention, and reducing operational inefficiencies, direct-to-patient logistics platforms are becoming critical to modern trial success.
As regulatory frameworks mature and digital integration deepens, the market is set to play a pivotal role in accelerating drug development timelines while ensuring patient safety and data integrity. This evolution positions clinical trial logistics as a cornerstone of next-generation healthcare innovation.
Explore More Related Studies Published by FMI Research:
Clinical Trials Market: https://www.futuremarketinsights.com/reports/clinical-trials-market
Clinical Trial Data Management Service Market: https://www.futuremarketinsights.com/reports/clinical-trial-data-management-service-market
Clinical Trial Biorepository & Archiving Solution Market: https://www.futuremarketinsights.com/reports/clinical-trial-biorepository-and-archiving-solutions-market
Clinical Trials Support Software Solutions Market: https://www.futuremarketinsights.com/reports/clinical-trials-support-software-solutions-market
Clinical Trial Packaging Market: https://www.futuremarketinsights.com/reports/clinical-trial-packaging-market
About Future Market Insights (FMI)
Future Market Insights (FMI) is a leading provider of market intelligence and consulting services, serving clients in over 150 countries. Headquartered in Delaware, USA, with a global delivery center in India and offices in the UK and UAE, FMI delivers actionable insights to businesses across industries including automotive, technology, consumer products, manufacturing, energy, and chemicals.
An ESOMAR-certified research organization, FMI provides custom and syndicated market reports and consulting services, supporting both Fortune 1,000 companies and SMEs. Its team of 300+ experienced analysts ensures credible, data-driven insights to help clients navigate global markets and identify growth opportunities.
For Press & Corporate Inquiries
Rahul Singh
AVP - Marketing and Growth Strategy
Future Market Insights, Inc.
+91 8600020075
For Sales - [email protected]
For Media - [email protected]
For web - https://www.futuremarketinsights.com/
SOURCE: Future Market Insights, Inc.
